Elena Cohort
Longitudinal study in children with autism

Overview
Developmental trajectories in Autism Spectrum Disorder (ASD) are highly variable, yet current knowledge remains limited to explain such diversity. This heterogeneity arises from many risk and protective factors — some of which can only be identified from large-scale samples. Cohort studies allow rigorous observation of change dynamics and developmental evolution profiles. However, internationally, large pediatric ASD cohorts remain rare and those existing are seldom conducted over long durations.
In this context was established the ELENA, cohort: a prospective, multicenter pediatric study, built on an interdisciplinary, multi-thematic and translational approach — with the goal of improving understanding of developmental trajectories in children with ASD.
Key Information
Start
2013
Main Objective
Developmental trajectories in children with ASD
Partenaires
Status
Phase 2 : data analysis
Key figures
2013
Launch of the ELENA Cohort
The first French multicenter and longitudinal cohort aiming to identify phenotypes and developmental trajectories of Autism Spectrum Disorder (ASD), along with their clinical, biological and environmental determinants.
Children aged between two and sixteen diagnosed with ASD, from 12 specialized centers across France.
Based on data collected within the ELENA cohort and making a significant contribution to the understanding of Autism Spectrum Disorder (ASD).
This is the duration of follow-up for participating families. Over these six years, five visits were organized to collect data through on-site assessments and online questionnaires.
Aiming to explore and deepen new crucial topics: gut microbiota, screening for NDD in siblings, care pathways, and environmental exposures.
In details
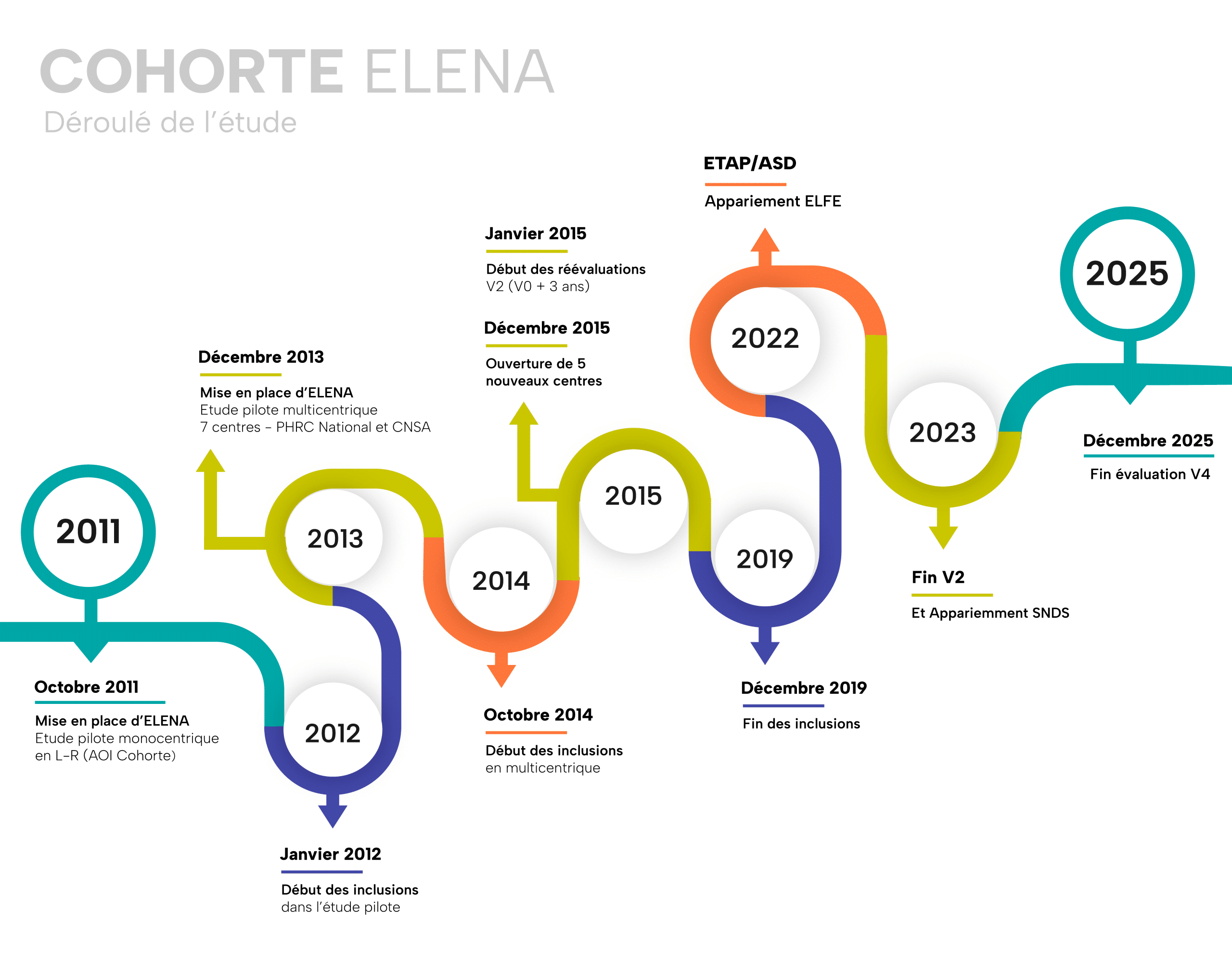
The ELENA cohort included 876 children under 16 years of age diagnosed at 12 specialized centers participating in the ELENA consortium. Each child is followed every 18 months for at least 36 months (Inclusion = V0, +18 months = V1, +36 months = V2, and +72 months = V4). The data collected cover clinical, medical, social, and environmental dimensions.
The primary outcome measure is adaptive social functioning, assessed using the Vineland II scale. Secondary outcomes include the severity of autistic symptoms, comorbidities, family quality of life, and the interventions received.
Recruitment was completed in December 2019, after six years of enrollment, and follow-up is scheduled to conclude in the first quarter of 2026.
Study Overview
Objectives
The main objective is to identify the developmental trajectories of a large population of children with ASD and to analyze the risk and protective factors to which these children are exposed throughout these trajectories
The secondary objectives are to assess the impact of ASD on family quality of life, to characterize the types of interventions offered and participants’ care pathways, to describe comorbidities and their evolution over time, and finally, to promote translational research by providing a reliable and as comprehensive as possible database on ASD.
The analysis of these results is expected to provide better answers to the many questions parents and professionals have regarding the development of each child and adolescent, to improve patient care, and to strucutre research so as to privide a public health tool that enhances understanding of population needs at different stages of life and improves professional practices.
Scientific Publications
Ancillary studies
Ancillary studies are supplementary studies to the initial research project. They aim to further investigate specific topics or to explore new research areas.
ETAP-ASD
Environmental exposures To Air Pollution and Autism Spectrum Disorder in children
ELENA-SNDS
Care Pathways of Children with Autism Spectrum Disorder Included in the ELENA Cohort: Linkage with the National Health Data System
EnviroDisorders
Environmental pesticides and genetic predisposition: a path toward autism spectrum disorders
FRATSA
Screening for Neurodevelopmental Disorders in Siblings of Children with Autism Diagnosed in Tertiary Centers
NUTRIL
Pilot Study on Early Maternal Breastfeeding in the First Year of Life and ASD Severity in the ELENA Cohort
LEDA
Study of the Link Between Gut Dysbiosis and Blood–Brain Barrier Integrity in Autism
ELENA-COVID
Study on the Psychological, Social, and Health Impact of the COVID-19 Pandemic in the ELENA Cohort
Data Access
Collaboration proposals related to the ELENA cohort are reviewed by the cohort steering committee.
To submit a scientific project proposal based on data from the ELENA cohort, please contact the ELENA team, who will be happy to support you throughout the process.
Contact
04.67.33.09.86
rech-clinique-autisme@chu-montpellier.fr
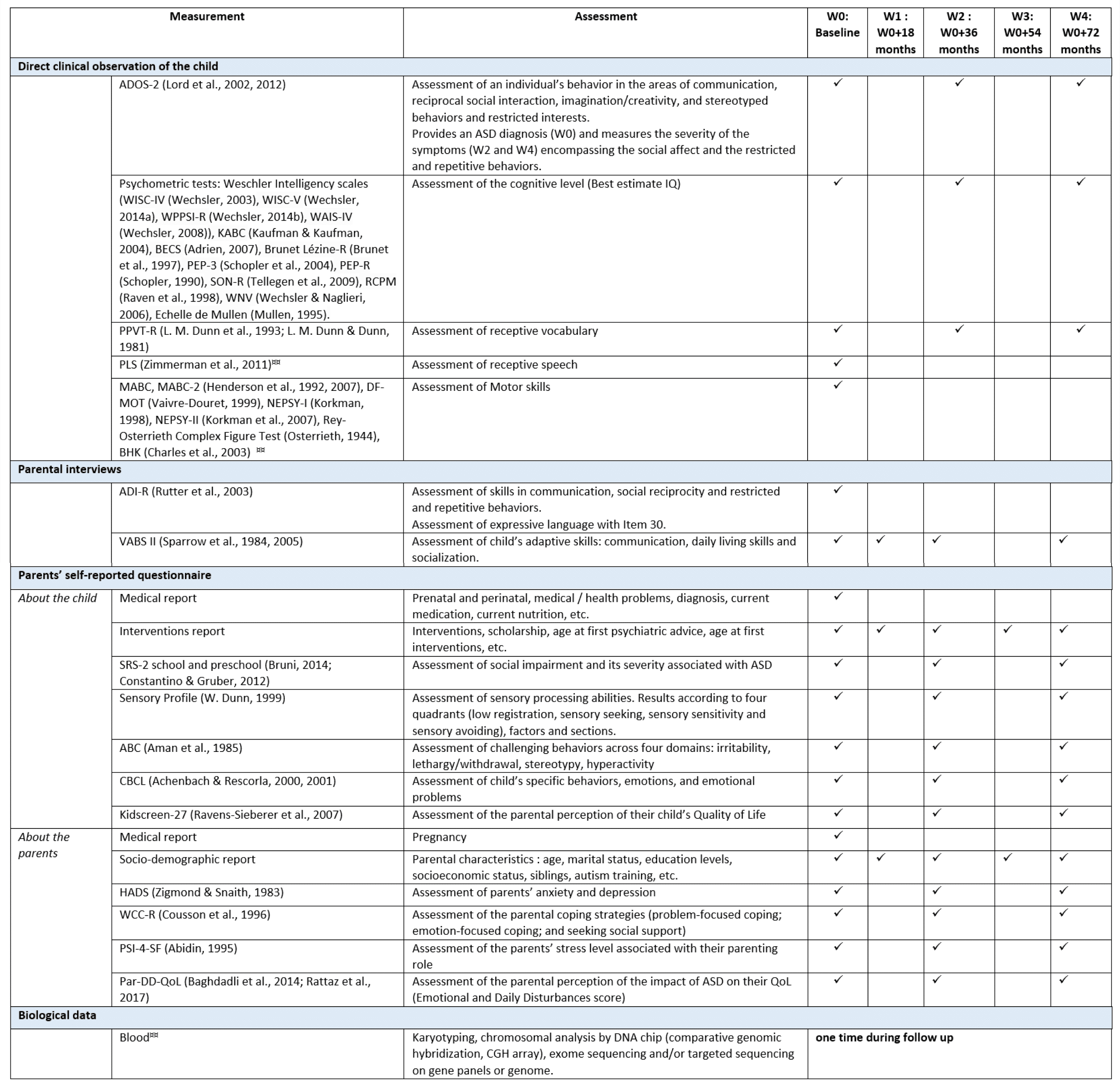
TABLE: Measures of the ELENA Study Protocol at Baseline and During Follow-up
ABC: Aberrant Behavior Checklist; ADI-R: revised version of the Autism Diagnostic Interview; ADOS: Autism Diagnostic Observation Schedule; ASD: autism spectrum disorder; BECS: Batterie d’Évaluation du développement Cognitif et Social; BHK: Concise Assessment Scale for Children’s Handwriting; CBCL: Child Behavior Checklist; CGH: Comparative genomic hybridization; DF-MOT: Motor Functional Development Scale; HADS: Hospital Anxiety and Depression Scale; KABC: Kaufman assessment battery for children; NEPSI: neuropsychological performance; Par-DD-QoL: Parental-Developmental Disorder-Quality of Life; PEP-R: Psychoeducational profile-revised; PEP-3: Psychoeducational profile-third edition; PPVT-R: Peabody Picture Vocabulary Test-Revised; PSI-4-SF: Parenting Stress Index, Fourth Edition Short Form; QoL: quality of life; RCPM: Raven’s Colored Progressive Matrices; SON-R: Revised Snijders-Oomen Nonverbal Intelligence Test SRS-2: Social Responsiveness Scale-2; VABS-II: Vineland Adaptive Behavior Scales, Second Edition; W: wave; WAIS: Wechsler adult intelligence scale; WISC: Wechsler intelligence scale for children; WCC: French Ways of Coping Checklist; WNV: Wechsler Nonverbal Scale of Ability; WPPSI : Wechsler Preschool and Primary Scale of Intelligence.
¤¤available only for the Montpellier Center.
Table adapted from the article: Baghdadli A, Peries M, Loubersac J, Michelon C, Rattaz C, Ferrando L, David A, Munir K, Picot MC. Contributions of the ELENA Cohort to Study Autism Spectrum Disorder in Children and Adolescents from a Biopsychosocial Framework. J Autism Dev Disord. 2024 Sep 4. doi: 10.1007/s10803-024-06519-8. Epub ahead of print. PMID: 39230780.
Organization
Coordinating Team
Baghdadli, A – Coordinateur Scientifique
Loubersac, J – Data Manager
Peries, M – Biostatistician
Esnee, C ; Gerbe C – Psychologists (Assessment)
Next
Picot, M-C – Epidemiologist & Methodologist
Michelon, C – Biostatistician
Ferrando, L – Clinical Research Coordinator
Rattaz, C ; Dellapiazza F – Psychologists (Dissemination)
Governance
Overall project governance is ensured by the medical coordinator (Prof. BAGHDADLI), a steering committee, and a scientific committee.
Resources for the General Public
Family Magazine - 2025 Edition

An annual magazine for families is prepared by the coordinating team of the ELENA Cohort. These newsletters are sent to all families. Their purpose is to inform families about the life and progress of the ELENA Cohort by providing a summary of the cohort, a current overview, and upcoming events.

Family Magazine – 2024 Edition
Discover the editorial by Prof. Amaria Baghdadli, interviews on motor and social skills in children with ASD, the role of a Clinical Research Officer, the FRATSA Project on screening for neurodevelopmental disorders, and much more.
Voices of Researchers
Regularly, a researcher explains to the general public the research findings published in scientific journals based on data from the ELENA cohort.
Anxiety in Children with Autism
Pesticides and Autism Spectrum Disorder in Children
Screen Exposure and Associated Risks in Children with Autism
Ethics and Confidentiality
This study, classified as standard care, received approval from the Sud Méditerranée I Ethics Committee (CPP), the Advisory Committee on the Processing of Information in Health Research (CCTIRS), and the French Data Protection Authority (CNIL). Although, from a regulatory standpoint, a standard care study only requires verbal consent from parents, we also systematically obtain their written consent.
Each parent remains completely free to withdraw at any time, for any reason. The same applies to children who, as they grow older, may wish to express their disagreement.
Anonymity is an absolute rule, but longitudinal follow-up requires that families be contacted regularly. We have therefore implemented a system comprising three separate databases. An administrative database contains the patients’ contact information and their anonymized ID numbers. This is the only database linking patient identities to their ID numbers and is accessible to only two team members responsible for contacting the families. The clinical and parental databases contain patient data in a fully anonymized form. Scientists will therefore always work with anonymized information.
In accordance with the provisions of the amended French Data Protection Act (Law No. 78-17 of January 6, 1978, on Information Technology, Data Files and Civil Liberties) and the General Data Protection Regulation (EU Regulation 2016/679), you have the following rights:
– The right to request access to, correction, deletion, or restriction of your data collected as part of the research;
– The right to object to the collection and transfer of your data protected by medical confidentiality;
– The right to object to the secondary use of your data and biological samples for conducting research, studies, and evaluations in the health field that serve the public interest within the scientific themes covered by the ELENA study;
– The right to withdraw your consent to the collection of your data at any time. However, please note that data already collected prior to the exercise of your right to object or to withdraw consent may continue to be processed confidentially so as not to compromise the achievement of the research objectives.
These rights can be exercised by contacting us via email at rech-clinique-autisme@chu-montpellier.fr